Introduction: The Hidden Science of Purity
When most people hear the words “water purification,” they picture a plastic pitcher filter or a small under-sink unit. For a pharmaceutical manufacturer, however, water is a raw ingredient. Impurities measured in parts per billion can ruin a life-saving vaccine or degrade a critical antibiotic.
This is where Commercial Reverse Osmosis Water Purification becomes the gold standard.
Unlike consumer-grade filters that merely improve taste, commercial systems are engineered to remove viruses, bacteria, dissolved salts, and endotoxins. At SKE&EAGLE, we design these high-stakes systems for the most regulated environments on earth. But what separates a standard industrial filter from a truly reliable machine? The answer lies in the materials, the validation, and the engineering behind the reverse osmosis process.
In this guide, we will explore the mechanics of modern RO systems, the critical role of Stainless Steel Reverse Osmosis System construction, and how to identify the Best Water Purification System for your production line.
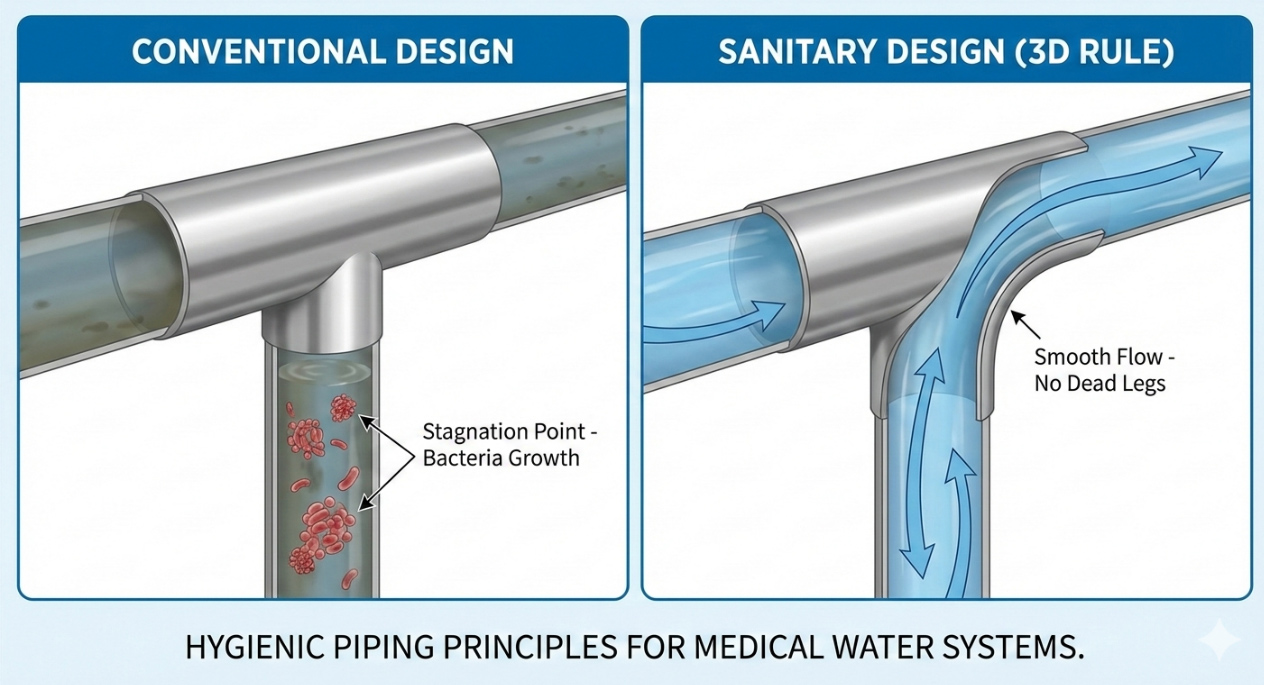
What is Commercial Reverse Osmosis Water Purification?
Reverse Osmosis (RO) is not a filtration method; it is a separation process. By applying pressure to a concentrated solution, you force water molecules through a semi-permeable membrane, leaving contaminants behind.
In a Commercial Reverse Osmosis Water Purification setup, this process happens on a massive scale. A typical system might process 500 to 50,000 gallons per day.
Here is the basic breakdown:
-
Pretreatment: Sediment filters and activated carbon remove chlorine and large particles (chlorine destroys RO membranes).
-
The RO Membrane: Water is forced through microscopic pores (0.0001 microns). Only H2O molecules pass through. Bacteria, pyrogens, and salts are rejected and flushed to the drain.
-
Post-Treatment: Depending on the industry, the “permeate” (purified water) may run through UV lights or deionization resins.
For pharmaceutical companies, this is only the first step. The purified water must then be stored in a loop to prevent biofilm growth.

The Material Advantage: Why Stainless Steel Matters
One of the biggest mistakes a facility can make is using plastic components in a high-purity water system. While plastic (like PVC or Polypro) is cheaper upfront, it is porous. Over time, these pores harbor bacteria, creating a biofilm that is impossible to fully eradicate without harsh chemicals.
This is why a Stainless Steel Reverse Osmosis System is non-negotiable in regulated industries.
Specifically, SKE&EAGLE fabricates systems using 316L Stainless Steel. Here is why this metal dominates the industry:
-
Passivation Layer: Stainless steel naturally forms a chromium oxide layer on its surface that is chemically inert. It doesn’t leach chemicals into your pure water.
-
Surface Finish (Ra < 0.6µm): We mechanically polish the interior surfaces to a mirror finish. With a roughness average below 0.6 micrometers, bacteria have nowhere to hide. They cannot attach, and they cannot proliferate.
-
Corrosion Resistance: Pharmaceutical sanitization often involves hot water (85°C) or ozone. Stainless steel handles these aggressive sanitizers without degrading.
If your goal is to produce Pure Water Reverse Osmosis quality (typically USP Grade), plastic piping will cause you to fail validation. Stainless steel is an investment in compliance.
Pure Water Reverse Osmosis: Defining the Standard
What exactly is “pure water”? In the commercial space, purity is measured by resistivity (MΩ·cm) or conductivity (µS/cm).
Pure Water Reverse Osmosis typically refers to water that has had 99% of all ionic and organic contaminants removed. However, a single-pass RO unit usually produces water at 10-50 µS/cm conductivity. This is pure enough for cooling towers or car washes, but not for injections.
To achieve “Highly Purified Water” (HPW) or Water for Injection (WFI), engineers use a double-pass RO or a combination of RO + Electrodeionization (RO/EDI).
The SKE&EAGLE Approach:
We integrate intelligent monitoring into every Commercial Reverse Osmosis Water Purification skid. Our systems include:
-
In-line conductivity meters: To ensure rejection rates stay above 99%.
-
Flow restrictors: To maintain the correct cross-flow velocity (self-cleaning the membrane).
-
Hot water sanitizable loops: For the downstream storage and distribution.
Without these features, the water coming out of the RO might look clean, but it will fail a simple microbial test.
How to Choose the Best Water Purification System
Searching for the Best Water Purification System online yields millions of results. However, “best” is contextual. The best system for a brewery (mineral content retention) is different from the best system for a lab (100% ion removal).
When evaluating vendors, use this checklist of industrial requirements:
| Feature | Consumer Grade | Commercial / Pharma Grade (SKE&EAGLE) |
|---|---|---|
| Material Construction | Plastic, Glass fiber | 316L Stainless Steel, Sanitary connections |
| Membrane Type | Thin-film composite (low pressure) | High-rejection, Low-energy (High pressure) |
| Sanitization | Chemical only (bleach) | Hot water, Steam, or Ozone capable |
| Data Integrity | None / Basic display | PLC with 21 CFR Part 11 compliance (Audit trails) |
| Validation | No documentation | IQ/OQ Protocols, Weld logs, Passivation reports |
The Best Water Purification System for a pharmaceutical plant is not the cheapest machine on Alibaba. It is the one that comes with Validation Documentation. SKE&EAGLE provides full Installation Qualification (IQ) and Operational Qualification (OQ) packages. This is the paperwork that proves your water system works to the FDA or EMA.

The Engineering Behind Stainless Steel Reverse Osmosis Skids
Let’s look inside a Stainless Steel Reverse Osmosis System designed by SKE&EAGLE.
Most people assume the tank is the most important part. Actually, the pump and the membrane housing are the heart.
-
The High-Pressure Pump: Built from 316SS to avoid rust particles entering the water stream. It pressurizes the feed water to 200-400 PSI.
-
The Housings: We use stainless steel pressure vessels to hold the spiral-wound RO membranes. Unlike fiberglass vessels, stainless allows us to sanitize the housing itself without cracking under thermal stress.
-
The Instrumentation: We install low-flow dead-legs. This means the pressure gauges and sensors are mounted via zero-static diaphragm seals so water does not sit stagnant in the sensor port.
Every weld on our Commercial Reverse Osmosis Water Purification skids is an orbital weld, documented via a weld log. We do not use manual stick welding, which leaves slag and gaps. We use orbital welding to create a continuous, seamless joint.
Maintenance and Longevity
Even the best stainless steel system requires a maintenance strategy. The most common failure point in Pure Water Reverse Osmosis systems is membrane fouling.
Scale: Calcium and magnesium precipitate on the membrane surface.
Biofouling: Slime builds up if chlorine levels drop.
Silica scaling: Extremely hard to remove; requires specific anti-scalants.
SKE&EAGLE’s recommendation for longevity:
-
Daily: Log pressure, flow rates, and conductivity.
-
Monthly: Flush the system if not in continuous use.
-
Quarterly: Chemical clean (CIP) using low-pH (acid) and high-pH (caustic) detergents.
-
Annually: Inspect stainless steel welds for pitting corrosion.
Because we use a Stainless Steel Reverse Osmosis System, our clients can use aggressive cleaning protocols that would destroy plastic systems. This extends membrane life from 2 years to 5+ years.
Conclusion: The SKE&EAGLE Difference
Selecting a water purification partner is a 10-year decision. You are not just buying a machine; you are buying compliance, uptime, and risk management.
SKE&EAGLE combines European engineering precision with the agility to build custom Commercial Reverse Osmosis Water Purification skids for the pharmaceutical and biotech industries. We refuse to cut corners. Every system features:
-
316L Stainless Steel wetted parts.
-
Sanitary tri-clamp fittings (no threads).
-
Automated hot water sanitization loops.
-
Full FAT (Factory Acceptance Testing) before shipping.
Whether you need a Pure Water Reverse Osmosis system for a small lab or a massive WFI generation plant for a vaccine facility, we deliver the Best Water Purification System for your specific water chemistry.

Frequently Asked Questions (FAQ)
Q1: Can a Commercial Reverse Osmosis Water Purification system remove viruses?
A: Yes. The pores in an RO membrane are approximately 0.0001 microns. Viruses typically range from 0.02 to 0.03 microns. Therefore, a properly maintained RO system provides a 4-6 log reduction of viruses. However, pharma standards usually require additional UV treatment as a redundant safety step.
Q2: Why is a Stainless Steel Reverse Osmosis System more expensive than plastic?
A: The cost difference comes from material prices and fabrication labor. Stainless steel requires skilled welders, argon back-purging, and electrochemical passivation. Plastic systems are glued or heat-fused. However, stainless steel lasts 30+ years in harsh environments, whereas plastic cracks, sags, and grows biofilm within 5-7 years.
Q3: How often do RO membranes need to be replaced?
A: For a Pure Water Reverse Osmosis system in a pharmaceutical environment (with proper pretreatment and sanitization), membranes typically last 3 to 5 years. SKE&EAGLE systems include pressure gauges to monitor the “Normalized Differential Pressure” – when this rises by 15%, it is time to clean or replace.
Q4: What is the difference between “Purified Water” (PW) and “Water For Injection” (WFI)?
A: Both require the same conductivity levels. The difference is microbial limits. WFI requires zero detectable endotoxins. While a Commercial Reverse Osmosis Water Purification system can produce PW, producing WFI usually requires adding a distillation step or a hot RO storage loop maintained at 80°C.
Q5: Does SKE&EAGLE provide validation documentation?
A: Absolutely. As a pharmaceutical equipment manufacturer, we provide a complete validation package including: Design Qualification (DQ), Installation Qualification (IQ), Operational Qualification (OQ), and weld logs. We cannot produce equipment for the FDA-regulated market without these documents.
Q6: Can I install a stainless steel system outdoors?
A: Yes, but with caveats. 316L stainless steel resists chloride corrosion, but we recommend a protective shelter or electrical heat tracing if the ambient temperature drops below freezing. SKE&EAGLE can supply outdoor-rated enclosures with IP55 protection.
Contact SKE & Eagle for Advanced Solutions
For customized system engineering solutions or integration of high-performance water treatment technologies, please contact SKE & Eagle. Our professional team collaborates closely with industrial partners to design, implement, and maintain solutions tailored to your operational needs.
Official Website: https://www.ske-eagle.com/
Contact Email: info@ske-eagle.com
Follow us on Facebook for the latest industry insights: SKE & Eagle Facebook
Business Inquiries: Visit our official website and submit the contact form at the bottom.

