Understanding Ultra Pure Water: Definitions and Key Characteristics
Core Parameters Defining Ultra Pure Water Quality
| Parameter | Ultra Pure Water Specification | Typical Measurement Unit | Regulatory Reference |
|---|---|---|---|
| Conductivity | ≤0.06 µS/cm (25°C) | MicroSiemens per centimeter | USP <643>, EP 2.2.1 |
| Resistivity | ≥18.2 MΩ·cm (25°C) | Megaohms centimeter | Gold standard for purity |
| Total Organic Carbon (TOC) | ≤0.5 ppb | Parts per billion | USP <643>, EP 2.2.44 |
| Microbial Load | ≤1 CFU/100 mL | Colony-forming units | USP <1231>, EP 2.6.12 |
| Endotoxins | ≤0.001 EU/mL | Endotoxin units | EP 2.6.14, WFI requirement |
| Particulates | ≤0.05 µm (≤90% removal) | Micrometers | ISO 14644-1 |
Distinction: Ultra Pure Water vs. Purified Water vs. Water for Injection (WFI)
- Purified Water: Used for non-sterile processes (e.g., equipment cleaning, non- injectable formulations). Meets USP/EP conductivity limits (≤1.3 µS/cm at 25°C) but no endotoxin control.
- Water for Injection (WFI): Required for sterile injectable drugs, biologics, and cell therapy. Must meet ultra pure water standards plus zero endotoxins. Produced via distillation or advanced filtration.
- Ultra Pure Water (UPW): The strictest standard, used in high-purity applications (e.g., bioprocessing, diagnostic manufacturing). Meets all UPW parameters, including sub-ppb TOC and near-absolute resistivity.

Core Technology: How an Ultra Pure Water Purification System Works
Stage 1: Pretreatment – Protecting Core Components
- Multi-Media Filtration: Removes sand, silt, rust, and colloids using quartz sand and filters. Reduces turbidity to ≤1 NTU, preventing membrane fouling.
- Activated Carbon Filtration: Adsorbs organic compounds, chlorine, and chloramines. Critical for protecting reverse osmosis (RO) membranes from oxidation—chlorine can degrade membrane material within hours.
- Water Softener: Removes calcium, magnesium, and other scale-forming ions to prevent RO membrane scaling, which reduces efficiency and shortens lifespan.
- Precision Filter (5 µm): Captures fine particles remaining after pretreatment, acting as a final barrier to protect high-pressure pumps and RO membranes.
Stage 2: Primary Purification – Reverse Osmosis (RO)
- How It Works: Raw water is pressurized and forced through a thin-film composite (TFC) RO membrane. Water molecules pass through the membrane, while ions, bacteria, and large organics are rejected.
- Dual-Stage RO Configuration: SKE&EAGLE uses dual-stage RO systems for maximum purity. The first stage removes bulk contaminants; the second stage polishes the water, reduces TOC, and lowers conductivity to ≤2 µS/cm.
- Key Benefits: Chemical-free purification, high rejection rates, and low energy consumption compared to traditional ion exchange.

Stage 3: Advanced Purification – Deionization (DI) and Electrodeionization (EDI)
- Deionization (DI): Uses ion exchange resins to capture remaining ions. DI systems can be mixed-bed (cation and anion resins combined) for high purity. However, DI resins require periodic regeneration with acids and bases, making them less ideal for continuous pharmaceutical operations.
Electrodeionization (EDI): A game-changing technology that combines ion exchange with an electric field. EDI continuously removes ions without chemical regeneration, producing stable ultra pure water (resistivity ≥18.2 MΩ·cm) 24/7.
- How It Works: Ions migrate through ion-exchange membranes toward the electrodes, where they are discharged. The electric field also inhibits microbial growth, reducing contamination risks.
- Advantages Over DI: No chemical waste, lower operational costs, and consistent water quality—critical for GMP compliance. SKE&EAGLE’s EDI modules are designed for pharmaceutical use, with built-in UV sterilization and microfiltration to ensure microbial control.
Stage 4: Polishing and Disinfection – Ensuring Long-Term Purity
- Ultraviolet (UV) Disinfection: Uses 185 nm and 254 nm UV light to destroy microbial DNA/RNA and break down organic compounds. 254 nm light inactivates bacteria and viruses, while 185 nm light oxidizes organics to reduce TOC.
- Microfiltration (0.2 µm): Removes any remaining microorganisms or particulate matter, ensuring the water is free of viable microbes at the point of use.
- Storage and Distribution: Ultra pure water is stored in 316L stainless steel tanks with closed-loop recirculation to prevent microbial growth. Tanks are equipped with breathing filters to avoid contamination, and circulation pumps maintain a flow rate of ≥1.0 m/s to minimize stagnation.

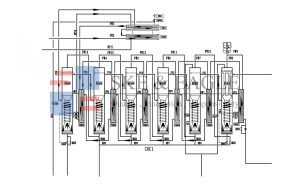
System Design and Components: SKE&EAGLE’s Engineering Excellence
Key System Components
| Component | Function | SKE&EAGLE Standard |
|---|---|---|
| High-Pressure Pump | Delivers pressure to RO membranes (15–25 bar) | Vertical multistage centrifugal pump with stainless steel construction |
| RO Membrane Skid | Houses dual-stage RO membranes | Filmtec or Hydranautics TFC membranes; 8-inch diameter for high flow |
| EDI Module | Produces ultra pure water (18.2 MΩ·cm) | Ionpure or Evoqua modules; integrated UV and microfiltration |
| Storage Tank | Stores ultra pure water | 316L stainless steel with insulation, breathing filter, and level sensor |
| Circulation Pump | Maintains closed-loop flow | Sanitary centrifugal pump with variable frequency drive (VFD) |
| UV Disinfection System | Inactivates microorganisms | Low-pressure UV lamp with automatic intensity control |
| Microfiltration Unit | Removes particulates and microbes | 0.2 µm PES or cellulose acetate membrane |
| PLC/HMI Control System | Automates operation and monitoring | Siemens or Allen-Bradley PLC; touchscreen HMI with real-time alerts |
| Sanitary Valves | Controls water flow | Tri-clamp diaphragm valves; stainless steel construction |
Design Principles for Pharmaceutical Excellence
- Redundancy: Critical components (pumps, membranes, EDI modules) are duplicated to ensure 24/7 operation. If one unit fails, the backup takes over without disrupting production.
- Energy Efficiency: SKE&EAGLE uses energy recovery devices (ERDs) to reduce power consumption by up to 40%, lowering operational costs.
- Compact Footprint: Skid-mounted systems are prefabricated in SKE&EAGLE’s facility, reducing installation time and site disruption. Ideal for facilities with limited space.
- Easy Maintenance: Modular design allows quick replacement of components (e.g., RO membranes, UV lamps) without system shutdown. Clear labeling and access panels simplify troubleshooting.
Applications of Ultra Pure Water Purification Systems
Pharmaceutical Manufacturing
- Injectable Drugs: Ultra pure water is used to formulate sterile injectables (e.g., vaccines, antibiotics, biologics). Any contamination could cause severe patient harm.
- Bioprocessing: For monoclonal antibodies, cell therapy, and gene therapy, ultra pure water is critical for cell culture, fermentation, and purification steps.
- Equipment Cleaning: GMP requires ultra pure water for cleaning sterile production equipment to avoid cross-contamination.
Medical Device Manufacturing
- Diagnostic Reagents: Reagents for blood tests, PCR, and immunoassays require ultra pure water to ensure accurate results.
- Sterile Devices: For devices like catheters and syringes, ultra pure water is used in cleaning and packaging processes.
Biotech and Life Sciences
- Cell Culture: Ultra pure water is essential for growing mammalian cells, as impurities can alter cell behavior or contaminate cultures.
- Research Laboratories: Analytical labs use ultra pure water for HPLC, mass spectrometry, and other high-precision tests.
Cosmetics and Personal Care
- High-End Formulations: Products like serums and creams require ultra pure water to avoid skin irritation and ensure product stability.
System Validation: Ensuring Long-Term Reliability
The Four Stages of Validation
- Design Qualification (DQ): Verifies the system design meets user requirements (URS) and regulatory standards. Includes review of process flow diagrams, component selection, and risk assessment (FMEA).
- Installation Qualification (IQ): Confirms the system is installed correctly per design specifications. Activities include verifying component certifications, piping integrity, and instrument calibration.
- Operational Qualification (OQ): Tests the system under controlled conditions to ensure it operates as intended. Includes performance testing (flow rate, pressure, TOC), alarm functionality, and sanitization validation.
- Performance Qualification (PQ): Validates the system under normal operating conditions over an extended period (30–90 days). Involves sampling at all points of use to ensure consistent water quality.
Post-Validation Maintenance
- Daily: Monitor conductivity, TOC, and pressure; log data; check for leaks.
- Weekly: Backwash pretreatment filters; inspect UV lamp intensity; clean sampling points.
- Monthly: Calibrate monitoring instruments; inspect EDI module performance; clean tank vents.
- Quarterly: Inspect RO membranes for fouling; replace precision filters; test sanitization effectiveness.
- Annually: Perform a full system audit; replace worn components (e.g., UV lamps, seals); update validation documentation.

Frequently Asked Questions (FAQs)
Q1: What is the difference between ultra pure water and purified water?
Q2: How often do RO membranes need to be replaced?
Q3: Can an ultra pure water system be customized for my facility?
Q4: What is the role of EDI in ultra pure water systems?
Q5: How long does it take to install and validate an ultra pure water system?
Conclusion
For customized system engineering solutions or integration of high-performance water treatment technologies, please contact SKE & Eagle. Our professional team collaborates closely with industrial partners to design, implement, and maintain solutions tailored to your operational needs.
Official Website: https://www.ske-eagle.com/
Contact Email: info@ske-eagle.com
Follow us on Facebook for the latest industry insights: SKE & Eagle Facebook
Business Inquiries: Visit our official website and submit the contact form at the bottom.

